Description
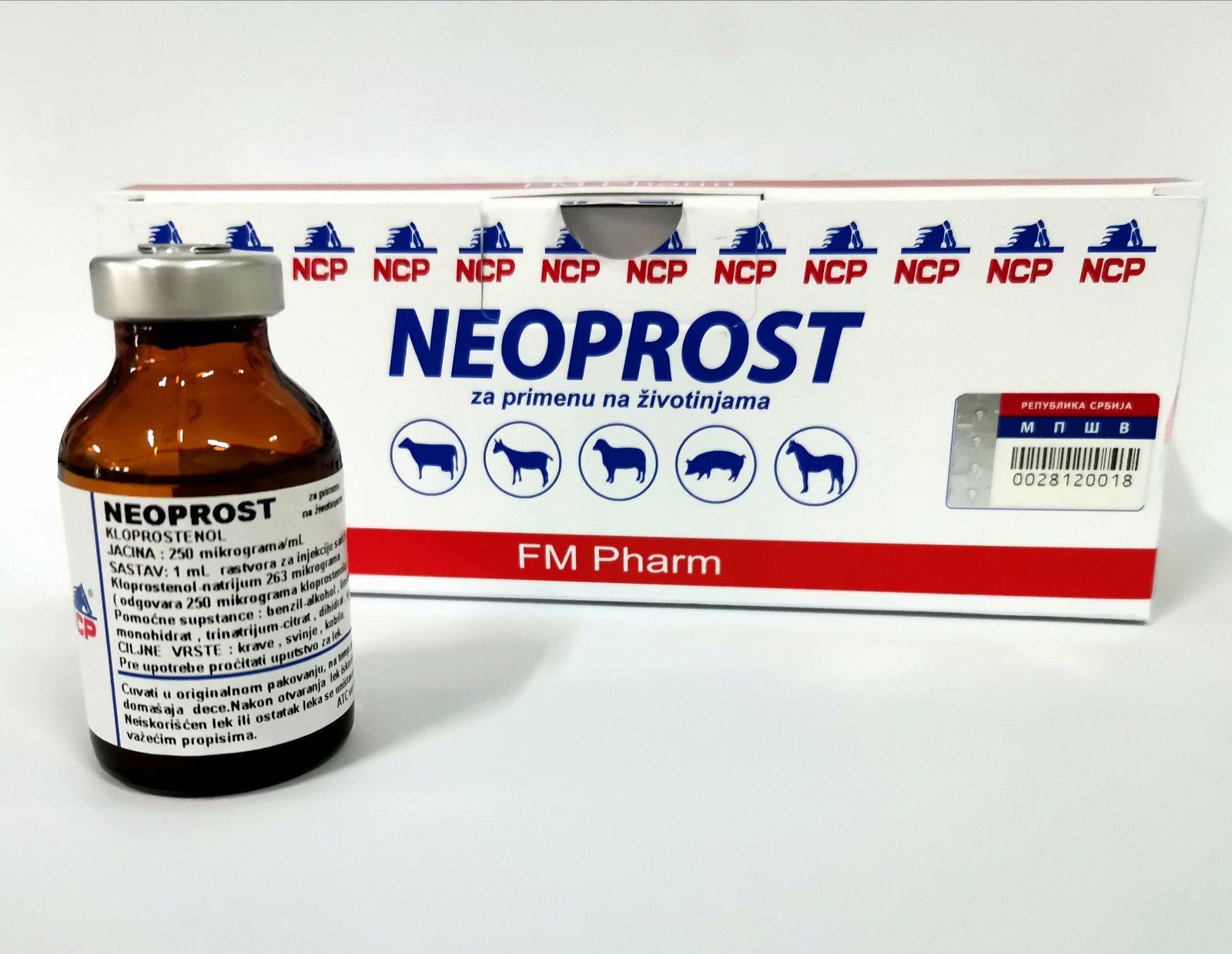
NEOPROST
QUALITATIVE AND QUANTITATIVE COMPOSITION OF THE MEDICINAL PRODUCT
Active substance:
Cloprostenol sodium 263 micrograms (equivalent to 250 micrograms of cloprostenol)
Excipients:
Benzyl alcohol
Citric acid, monohydrate
Trisodium citrate, dihydrate
Water for injections
INDICATIONS
Cattle (heifers, cows):
• To schedule the time of oestrous and ovulation and for cycle synchronization in animals with an ovulatory cycle when applied during the diestrus (induction of oestrus in non-detected oestrus, synchronisation of oestrus)
• Treatment of anoestrus and uterine disorders caused by a progesteroneinduced oestrous cycle blockade (induction of oestrous in anoestrus, endometritis, pyometra, corpus luteal cysts, follicular luteal cysts, shortening of the sexual rest period)
• Induction of abortion up to day 150 of pregnancy
• Expulsion of mummified foetuses
• Induction of parturition
Pigs (sows):
• Induction of farrowing
Mares:
• Estrus induction and synchronization (use only in case of previously performed ultrasound diagnosis of functionalcorpus luteum)
• Supportive therapy for pyometra and endometritis
CONTRAINDICATIONS
The drug is not given to pregnant animals unless pregnancy is interrupted. It is not recommended to administer sows if difficult dusting is expected due to the pathological position of the fetus or mechanical obstruction of the birth canal. It is contraindicated in animals with cardiovascular and respiratory diseases.
ADVERSE ACTIONS
Anaerobic infections may occur if anaerobic bacteria are introduced into the tissue by the injection, in particular following intramuscular injection.
Cattle
When used for induction of parturition, the incidence of retained placenta may be increased
depending on the time of treatment.
Mares
Sweating, increased respiratory and heart rates, ataxia, watery diarrhoea and signs of mild abdominal
pain in mares.
TARGET SPECIES
Cattle (heifers, cows), pigs (sows) and mares
DOSAGE AND METHOD OF ADMINISTRATION
The drug is administered via intramuscular injections.
Cattle (heifers, cows): 0.5 mg Cloprostenol/animal corresponding to 2.0 ml of the product /animal
-Induction of oestrus: (and in cows with poorly expressed or masked oestrus) the drug should be given after confirming the presence of the corpus luteum (days 6 to 18 of the cycle). Estrus usually occurs between 48-60 hours after drug administration. Insemination should be performed 72-96 hours after drug administration. If the animal does not enter the oestrus, repeat administration after 11 days.
-Induction of parturition : A drug given after 270 days of pregnancy. Expected delivery occurs 30-60 hours after drug administration.
-Estrus synchronization: give the drug twice (at 11 days intervals). Conduct artificial insemination 72 to 96 hours after the second application.
-Ovarian dysfunction: the drug should be given after the presence of the corpus luteum is established and inseminated in the first oestrus after administration of the drug. If the animal does not enter the oestrus, perform a gynecological examination again and resume therapy 11 days after the first application. Inseminate 72-96 hours after drug administration.
-Endometritis or pyometra: give one dose of medicinal product. Repeat the dose 10-11 days later, if necessary.
-Interruption of pregnancy: drug given during the first half of pregnancy.
-Embrional mummification: give one dose. The fruit will be harvested after 3 to 4 days.
-Slow uterine involution: give a single dose and, if necessary, repeat the drug once or twice at intervals of 24 hours.
Sows:
The dose for sows is 250 mcg of cloprostenol per animal, which is equivalent to 1 mL per animal.
Inductionof farrowing : The drug is given after 112 days of gestation.
Repeat after 6 hours.
Alternatively, a myometrial stimulator (oxytocin or carazolol) may be administered 20 hours after the first dose of d-cloprostenol. If a dual administration protocol is used, in about 70% of cases,
the birth occurs 20-30 hours after the first administration of the drug.
Mares:
The mares are given a single dose of 1ml (equivalent to 250 mcg of cloprostenol).
INSTRUCTIONS FOR CORRECT ADMINISTRATION
Do not use for intravenous administration.
To reduce the risk of anaerobic infections care should be taken to avoid injection through contaminated areas of skin. Clean and disinfect injection sites thoroughly before application.
WITHDRAWAL
Meat and edible tissues: 1 days
Milk: 0 days
It is not used in mares whose meat is intended for human consumption.
SPECIAL WARNINGS FOR THE STORAGE OF THE MEDICINAL PRODUCT
Store at the temperature 25°C, in original packaging.
SPECIAL WARNINGS
Do not use in pregnant animals when abortion or induction of parturition is not intended. Safety of the product has not been established during lactation.
Do not use in animals being treated with non-steroidal anti-inflammatories, as the synthesis of endogenous prostaglandins is inhibited.
This product must be handled carefully to avoid accidental self-injection or contact with the skin or mucous membranes of the user.
Prostaglandins of the F2α type may be absorbed through the skin and may cause bronchospasm or miscarriage.
Pregnant women, women in childbearing age, asthmatics and people with other respiratory tract diseases should wear waterproof gloves during administration of the product.
Accidental spillage on the skin should be washed off immediately with soap and water.
In case of accidental self-injection, seek medical advice immediately and show the package leaflet or the label to the physician.

Reviews
There are no reviews yet.